Cytokeratin 18 (CK18 and ccK18)
Non-invasive biomarkers for assessing hepatocyte apoptosis and necrosis
| Biomarker | Utility in NASH Studies | Available Assays | Catalog # |
| Total CK18 (M65®) | Biomarker for hepatocyte cell death (apoptosis and necrosis) | M65® ELISA | P10020 |
| M65® EpiDeath ELISA | P10040 | ||
| caspase-cleaved CK18 (ccK18/M30®) | Biomarker for hepatocyte apoptosis | M30® Apoptosense ELISA | P10011 |
The Problem:
In early-phase NASH trials, performing liver biopsies to evaluate the effect of a potential drug candidate is not always practical or feasible.
- These relatively short-duration early trials may not result in significant changes in liver histology.
- Traditional non-invasive biomarkers for NASH, such as serum ALT and AST levels, are notoriously insensitive and non-specific, and do not always accurately reflect the extent of liver injury.
The Solution:
Non-invasive, mechanistic biomarkers that are reflective and predictive of changes in liver histology
- Serum CK18 (M65®) and ccK18 (M30®) measure hepatocyte apoptosis and necrosis.
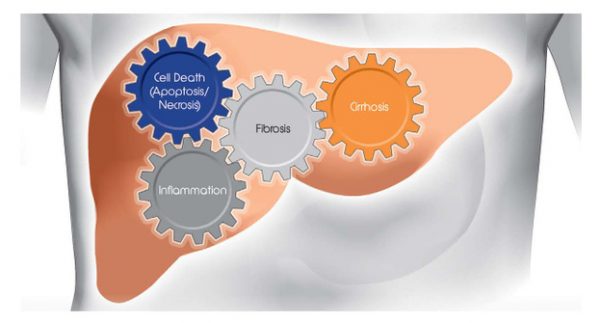
- Apoptosis and necrosis are underlying mechanisms that are directly involved in the pathogenesis of NAFLD
- Changes in CK18 and ccK18 levels may therefore be early indicators for changes in disease activity in response to drug treatment.
- Changes in serum CK18 and ccK18 levels have been shown to correlate with histological changes in the liver.
Additional NASH Biomarker ELISAs from DiaPharma
DiaPharma offers additional non-invasive biomarker assays for NAFLD/NASH.
Incorporating these biomarkers into your research adds valuable information about changes in disease activity, which could decrease the time and costs of studies.
- M30® and M65® Cytokeratin 18 (CK18)ELISAs
- Osteopontin
- Mac-2 Binding Protein
- sCD163
- L-FABP
- FGF-19
- FGF-21
For research use only in the U.S. and Canada. Not for use in diagnostic procedures.