Antithrombin (AT, Antithrombin III, ATIII) is a small glycoprotein produced by the liver that inactivates several enzymes of the coagulation system.
α-Antithrombin is the dominant form of antithrombin found in blood plasma and has an oligosaccharide occupying each of its four glycosylation sites. A single glycosylation site remains consistently un-occupied in the minor form of antithrombin, β-antithrombin.
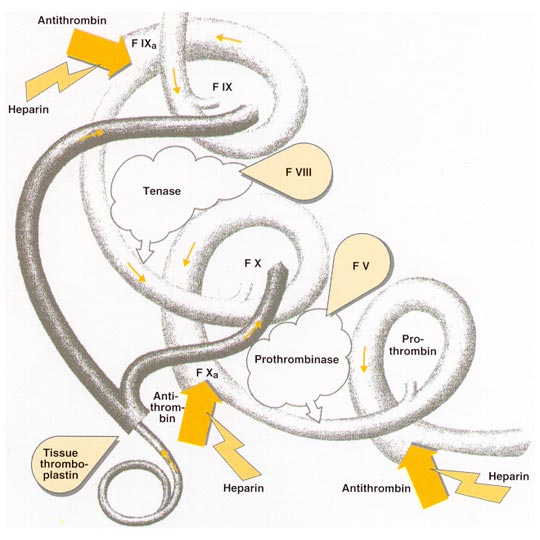
Antithrombin activity is increased by anticoagulant heparin, which enhances the binding of antithrombin to factor II and factor X.
Antithrombin is a serpin (serine protease inhibitor) similar in structure to other plasma protease inhibitors like alpha 1-antichymotrypsin, alpha 2-antiplasmin and Heparin cofactor II.
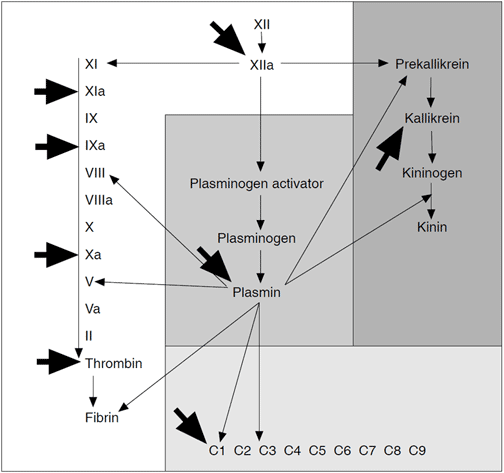
Antithrombin targets proteases on the contact activation pathway (intrinsic pathway), the activated forms of Factor X (Xa), Factor IX (IXa), Factor XI (XIa), Factor XII (XIIa), Factor II (thrombin) (IIa), and Factor VII (VIIa) from the tissue factor pathway (extrinsic pathway). The inhibitor also inactivates kallikrein and plasmin, also involved in blood coagulation. It inactivates serine proteases that are not involved in coagulation such as trypsin and the C1s subunit of the enzyme C1 involved in the complement pathway.
Antithrombin deficiency – A Risk Factor for Venous Thrombosis
Introduction
Antithrombin is a circulating plasma protein that functions as an important regulator of blood coagulation. It inactivates several enzymes of the coagulation cascade, in particular thrombin and factor Xa. Since a link between hereditary antithrombin deficiency and thrombosis was established in 1965, there has been increasing clinical interest in antithrombin and a need for simple and accurate determination. Assays based on chromogenic peptide substrates are now available, allowing photometric detection of antithrombin activity in plasma.
Thrombin is the key enzyme in blood coagulation. It clots blood by converting fibrinogen into clot-forming fibrin monomers and activates factor XIII leading to the strengthening of the blood clot by cross-linking. Thrombin also activates platelets and the cofactors, factor V and factor VIII, thereby accelerating its own generation and providing a quick response to injury. However, the autocatalytic nature of thrombin also entails certain hazards. The complete thrombin generation of only one millilitre of plasma would, unless regulated, clot the blood content of an adult within minutes. It is thus apparent that thrombin activity must be closely controlled in order to prevent abnormal fibrin deposition within the vasculature. Inhibitory regulation of thrombin is primarily achieved by two principally different mechanisms. When thrombin binds to the membrane protein thrombomodulin present at the surface of intact vascular endothelium, it loses all of its procoagulant properties. Instead, thrombomodulin dramatically accelerates the rate at which thrombin activates protein C. Activated protein C then degrades factors Va and VIIIa, effectively impeding further thrombin generation.
The other regulatory mechanism of thrombin activity generated during blood clotting is provided by a group of circulating enzyme inhibitors. Antithrombin (also called antithrombin III) is the major inhibitor, accounting for approximately 80% of the thrombin inhibitory activity in plasma.
Human antithrombin data
| Name: | Antithrombin |
| Synonyms: | AT, antithrombin III, ATIII, heparin cofactor I, thrombin inhibitor I |
| History: | Discovered by Morawitz in 1905 |
| Plasma concentration: | 150 mg/l |
| Molecular weight: | 58,200 daltons |
| Primary structure: | 432 amino acid residues |
| Carbohydrate content: | 15% |
| Biological half-life: | 55-70 h |
| Gene: | 13.5 kb, 7 exons, 6 introns location chromosome 1 at 1q 23-25 |
| Type: | Belongs to a superfamily of about 40 serine protease inhibitors (serpins) which have diverged from a common ancestral molecule. |
| Function: | Inhibits thrombin, factor Xa and other proteases. Cofactor for heparin. |
| Importance: | Antithrombin deficiency is associated with an increased risk of developing a thromboembolic disorder |
Thrombin Inhibitors in Plasma
| Inhibitors | Target protease(s) |
| Antithrombin | Thrombin, IXa, Xa, others |
| a2-macroglobulin | Non-specific |
| Trypsin inhibitor | Neutrophil elastase, Thrombin, others |
| Heparin cofactor II | Thrombin |
The Coagulation Cascade
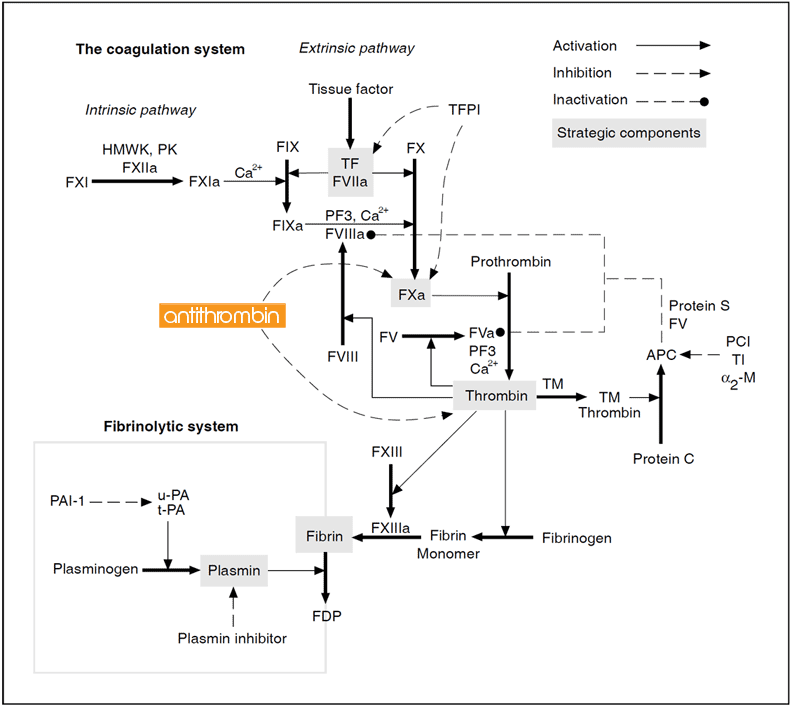
Blood coagulation is initiated by vascular injury and results in the explosive generation of thrombin which clots blood. Coagulation factors are represented by their Roman numerals (a = activated). Abbreviations: HMWK= high molecular weight kininogen, PK = prekallikrein, K = kallikrein, TF = tissue factor, TFPI = tissue factor pathway inhibitor, PF3 = phospholipid, TM = thrombomodulin, PC = protein C, APC = activated protein C, PCI = protein C inhibitor, TI = trypsin inhibitor, a2-M = a2-macroglobulin, FDP = fibrin degradation products. Notes: Factor V has recently been identified as a second APC cofactor.
Plasma coagulation factors and regulatory proteins
| Factor | Name | Size [kDa] | Concentration [mg/ml] |
| I | Fibrinogen | 340 | 3000 |
| II | Prothrombin | 69 | 100 |
| III | Tissue factor | 47 | – |
| IV | Calcium | – | – |
| V | Proaccelerin | 330 | 10 |
| VI | – | – | – |
| VII | Proconvertin | 48 | 0.5 |
| VIII | Antihemophilic factor | 330 | 0.1 |
| IX | Christmas factor | 55 | 5 |
| X | Stuart-Prower factor | 59 | 8 |
| XI | Thromboplastin antecedent | 160 | 5 |
| XII | Hageman factor | 80 | 30 |
| XIII | Fibrin-stabilizing | 320 | 10 |
| – | Tissue factor | 37 | – |
| – | Protein C | 57 | 4 |
| – | Protein S | 75 | 25 |
| – | Antithrombin | 58 | 150 |
| – | Heparin cofactor II | 66 | 91 |
Biochemistry of Antithrombin
Structure
Antithrombin is a 58 kDa single-chain plasma glycoprotein and a member of the serpin family of serine protease inhibitors. It is synthesized in the liver and occurs in normal plasma at a concentration of about 150 mg/l, with a half-life of 3 days. The primary structure of human antithrombin in plasma comprises 432 amino acid residues, six of which are cysteine that form three intermolecular disulfide bonds. The molecule also contains four carbohydrate side chains which make up around 15% of the molecular mass.
Function
Antithrombin is the major inhibitor of thrombin, factor IXa, and factor Xa in plasma, but it also inactivates the other serine proteases of the intrinsic coagulation pathway, factors XIa and XIIa, as well as some noncoagulation serine proteases, such as plasmin, kallikrein and the complement enzyme C1. Most proteases are inactivated much more slowly than thrombin. Inhibition by antithrombin involves the formation of a stable 1:1 complex between the active domain of the serine protease and the reactive site of antithrombin, which proteases initially recognize as a substrate. During the cleavage of the reactive site bond in antithrombin (located at Arg393-Ser394), a conformational change occurs in the inhibitor that traps the protease.
Interactions with heparin
Protease-antithrombin interactions are slow, although enhanced dramatically in the presence of certain sulfated polysaccharides known as glycosaminoglycans (GAGs). It is believed that vascular GAGs, primarily heparan sulfate, bind antithrombin and thrombin and thereby catalyze the antithrombin-thrombin reaction. This would permit the selective enhancement of antithrombin actions at blood-cell interfaces, where the coagulation enzymes are generated.
Commercial heparin is a mixture of GAGs extracted mainly from bovine or porcine intestinal mucosa and used as an important antithrombotic drug. Both heparin and heparan sulfate catalyze the actions of antithrombin by inducing a conformational change in the antithrombin molecule at the reactive site.
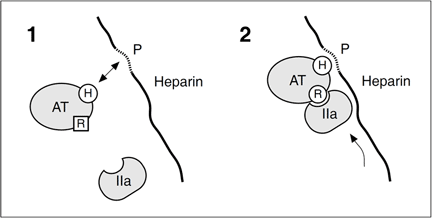
A simplified model describing how heparin catalyzes the formation of an irreversible complex between thrombin (IIa) and antithrombin (AT). [H] symbolizes the heparin binding site and [R] is the reactive site in antithrombin, normally in an unfavorable conformation for protease inhibition.[P] is the unique antithrombin binding segment of heparin. Binding to [P] induces a conformational change in antithrombin that facilitates its reaction with thrombin. Thrombin binds to heparin in a non-specific manner and ‘slides’ along the chain until it encounters the bound antithrombin. The affinity of heparin to the thrombin-antithrombin (TAT) complex is much lower than to free antithrombin. Heparin will therefore dissociate from the TAT complex, which is rapidly removed from the circulation by receptors in the liver (t1/2< 5 min). Factor Xa inactivation is also enhanced by heparin, although the sliding mechanism is not relevant for this enzyme as only the conformation change is required.
Antithrombin deficiency
Thrombophilia
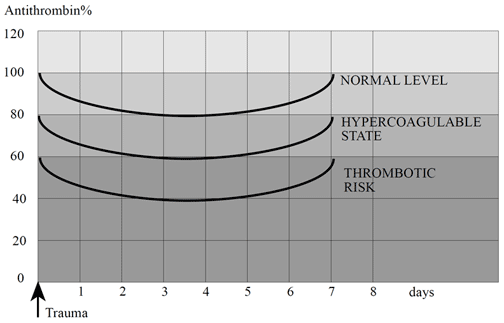
Venous thrombosis is a serious medical problem that annually affects 1 in 1000 individuals, causing considerable morbidity and death due to pulmonary embolism. The pathogenesis of the disease is multifactoral and several circumstantial as well as genetic risk factors have been identified.
Thrombophilia is the term used for familial or acquired defects of the hemostatic mechanism that increase the tendency to clot. The term encompasses a susceptibility to both venous and arterial thrombosis although, in the majority of patients, venous thrombosis of the lower limbs is the predominant clinical problem. Deficiencies of antithrombin, protein C, protein S, and the recently discovered inherited resistance to activated protein C (APCresistance), are the most common known genetic causes for thrombosis. Together, they constitute about 60-70% of cases diagnosed as inherited thrombophilia and may present as predisposing factors in up to one third of all affected patients with thrombosis.
The laboratory diagnosis of these patients is of utmost important, since it may help determine the type and the duration of treatment.
Clinical Aspects of Antithrombin
Hereditary antithrombin deficiency
Hereditary antithrombin deficiency was the first recognized inherited thrombophilia. It was reported in 1965 by Egeberg, who described a Norwegian family with recurrent episodes of venous thromboembolism.
Since then, families with hereditary antithrombin deficiency have been found in many countries and the link between antithrombin deficiency and thrombosis has now been clearly established. The pattern of inheritance is usually autosomal dominant (non-sex linked), with levels of functionally active protein around 40-70% of normal.
Prevalence
The prevalence of symptomatic hereditary antithrombin deficiency is estimated at between 1 in 2,000 and 1 in 5,000 in the general population, although the total prevalence, i.e. symptomatic and asymptomatic individuals, may be much higher. Recent studies of healthy blood donors estimate the incidence of hereditary antithrombin deficiency to be between 1 in 250 and 1 in 500.
Up to 5% of patients with a history of venous thrombosis, presenting with thrombosis at an age lower than 40-45 years, have a hereditary antithrombin deficiency. In an unselected group of patients with acute venous thrombosis the prevalence is about 1%.
Classification
Various mutations causing hereditary antithrombin deficiency have been found and classified into a number of different types. The most common scheme divides antithrombin deficiency into two types based on the results from functional and immunological assays. Type I antithrombin deficiency is the “classic” form of disorder and is characterized by a 50% reduction in both antigen and functional activity levels. Type II deficiency covers those cases in which approximately half the plasma antithrombin is a variant protein with reduced activity. Type II deficiency is subdivided into: mutations affecting the reactive site (RS), mutations affecting the heparin binding site (HBS), and pleiotropic (multiple) effects (PE). A more precise classification scheme based on the molecular defects has recently been proposed.
Risks and clinical features
The risk of a thromboembolic event in symptomatic antithrombin deficient individuals is on average approximately 1% per year of life. However, the risk appears to be age-related with the peak incidence occurring between the ages of 15 and 40.
About one-third of thrombotic episodes occur spontaneously, while in the remaining two-thirds identifiable risk factors include pregnancy or childbirth, prolonged immobilization, oral contraceptives containing oestrogen and major trauma.
The most common presentation of antithrombin deficiency is venous thrombosis of the lower limbs. A history of recurrent thrombosis occurs in about 60% of patients and is the clinical feature that usually prompts a search for antithrombin deficiency.
The clinical features of hereditary antithrombin deficiency
- Thrombotic episode occuring at an early age
- A family history of venous thrombosis
- Recurrent venous thrombosis
- A thromboembolic event at an unusual site
- Thrombosis during pregnancy
- Heparin resistance
Classification, prevalence and thrombosis risk of inherited antithrombin deficiency.
| Classification | Activity | Antigen | Prevalence* | Thrombosis risk** |
| Type I | Low | Low | 1:4200 | High |
| Type II | Low | Normal | 1:280 | High (RS), Low (HBS) |
| * The prevalence in 4,200 blood donors in the West of Scotland.5 ** Type I deficiency is associated with a high risk of recurrent thromboembolic disease, whereas the risk associated with type II deficiency depends on the location of the mutation. Individuals with mutations at or close to the reactive site (RS) have a high risk of thrombosis. In contrast, heterozygous individuals with mutations affecting the heparin-binding site (HBS) run a relatively low risk. | ||||
Acquired antithrombin deficiency
Acquired antithrombin deficiency is due to either decreased synthesis (e.g. liver disease, premature neonates), increased loss (e.g. nephrotic syndrome), increased consumption (e.g. DIC), or is drug-induced (e.g. heparin, L-asparaginase). Because acquired antithrombin deficiency is usually accompanied by decreases in other coagulation proteins, it is difficult to estimate the thrombotic risk based on antithrombin alone. However, in some cases the acquired antithrombin deficient condition entails a similar risk of thrombosis as the hereditary state and early recognition is therefore important.
DIC syndrome
A number of primary events such as trauma, shock, burns, infection, cancer, complicated pregnancy (pre-eclampsia), may cause a loosely-defined syndrome called disseminated intravascular coagulation (DIC). The syndrome is characterized by poorly controlled protease activity, resulting in accelerated fibrin formation and lysis which ultimately causes multiple organ damage. This happens when large amounts of traumatized or necrotizing tissue in the body releases tissue factor into the blood. Laboratory indications include a prolonged APTT, thrombocytopenia, hypofibrinogenemia and increased fibrin and fibrinogen degradation products. Finding a low level of antithrombin also suggests DIC, particulary if this value declines over time. In acute DIC, the consumption of antithrombin greatly exceeds the rate of synthesis and the body is unable to replace antithrombin quickly enough. In these cases the antithrombin level is useful as a diagnostic aid and for the evaluation of therapy. A severe initial decrease in antithrombin levels is a strong predictor of death and multiple organ failure in patients with septic DIC. Neonate antithrombin levels Antithrombin activity in healthy term neonates is approximately 50% of adult levels, with an increase to the normal range about 6 months after birth. The inhibitor a2-macroglobulin plays a more important role during this time, which partially explains why thrombosis is uncommon in healthy neonates. Plasma levels of antithrombin are about 35% in healthy preterm infants but may be decreased further in complications such as respiratory distress, sepsis or DIC. Sick infants with very low antithrombin levels have been reported to have a higher mortality rate, with a higher incidence of thrombotic and hemorrhagic complications.
Drug-induced effects
Heparin therapy decreases plasma antithrombin levels by increasing the hepatic clearance of antithrombin. In some cases a phenomenon called heparin resistance may develop when antithrombin levels are critically low.
Conditions that can lead to an acquired reduction in antithrombin levels
Physiological deficiency
- Pre-term infants and neonates
- Old age
Consumption coagulopathies
- DIC
- Pre-eclampsia
- Surgical procedures and post-operative period
- Acute myeloid leukemia
- Massive venous thrombosis
- Sick, pre-term infants and neonates
Renal disease
- Nephrotic syndrome
- Hemolytic uremic syndrome
Hepatic disease
- Acute liver failure
- Chronic liver disease
Gastrointestinal disease
- Inflammatory bowel
- Protein-losing enteropathy
Drugs
- Heparin
- L-Asparaginase
- Oestrogens, tamoxifen
Dilutional effects
- Hemodialysis
- Plasmapheresis
- Cardiopulmonary bypass
Other conditions
- Diabetes
- Behçet’s disease
- Malnutrition
Management of antithrombin deficiency
Antithrombin levels
A normal antithrombin range is assumed to be 80-120%,23 although there is a difficulty in knowing precisely what constitutes the lower limit of the normal range and what is the significance of levels just above or around this limit. Because of this, investigation of antithrombin must always include laboratory results as well as the patient’s clinical history. Higher concentrations than normal appear to have no clinical significance.
Prophylaxis and treatment options
The role of prophylaxis for individuals with hereditary antithrombin deficiency is at present unclear and controversial. However, it is agreed that individuals with symptomatic hereditary antithrombin deficiency should be offered prophylactic anticoagulants in situations that present a recognized risk of thrombosis (e.g. pregnancy, immobilisation and surgery). Attempts should be made to classify the type of deficiency in both symptomatic and asymptomatic individuals, in order to properly assess the risk of thrombosis and the need for prophylaxis. In general, patients with antithrombin deficiency type I and type II (RS) run a high risk of thrombosis.
Heparin
Heparin is the first line of treatment for acute thrombosis in antithrombin deficient patients. Heparin resistance and extension of thrombosis have been reported although in general this is not a major problem. It is probably not cost-effective to give antithrombin substitution to patients requiring heparin, in order to overcome presumed heparin resistance.
Warfarin
In the absence of contra-indications (e.g. hemorrhagic diathesis, severe hypertension, pregnancy) the choice of a long-term prophylactic is the oral anticoagulant warfarin. Warfarin is a vitamin K antagonist
and interferes in the vitamin K-dependent biosynthesis of several clotting factors by reducing their procoagulant activity.
Antithrombin concentrates
Although plasma may be used as a source of antithrombin, the large volume required for therapeutic replacement represents a major limitation. Unlike plasma, a pure antithrombin concentrate can restore balance to an unbalanced system. Antithrombin concentrates are usually prepared by affinity chromatography and are carefully treated to destroy infectious agents.
The successful use of antithrombin concentrates as prophylactics during surgical procedures or in the management of various consumptive coagulopathies have been reported by several groups. However, the definitive advantage of using antithrombin concentrates has still not been adequately proven.
Because of the moderate survival time of antithrombin in the blood circulation, alternate day infusions of antithrombin concentrates are usually necessary. Plasma levels should be monitored regularly to achieve effective treatment at a reasonable cost.
Antithrombin levels in various clinical states
| Condition | Range | Ref. |
| Liver disease | 25- 135% | 25 |
| DIC | 31-137% | 26 |
| Heparin* | 40-78% | 32 |
| Normal | 84-116% | 23 |
| Healthy neonates | 39-87% | 28 |
| Hereditary def. | 40-70% | 19 |
| *Continuous infusion of therapeutical dose | ||
Antithrombin Assay Methods
Antithrombin assays
Diagnosis of antithrombin deficiency
The diagnosis of antithrombin deficiency is primarily based on the determination of antithrombin levels in plasma by using a functional activity assay or an immunological assay. For screening purposes the functional antithrombin assays are most relevant since they can detect both types of hereditary antithrombin deficiencies.
Activity levels are usually expressed relative to a normal plasma pool with the designated value of 100%, which is equal to one unit of antithrombin in one millitre of reference plasma.
Activity assays
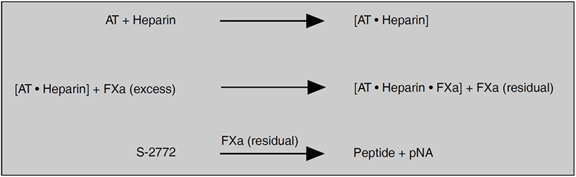
Functional assays are based on the capacity of a plasma sample to inhibit a known amount of either thrombin or factor Xa added in excess, in the presence of heparin (heparin cofactor assay) or in the absence of heparin (progressive activity assay). The residual enzyme is measured by a clotting-based assay or by using chromogenic peptide substrates.
Heparin cofactor assays
In the absence of heparin, the inhibition rates are slow and progressive antithrombin assays are influenced by other plasma inhibitors such as trypsin inhibitor and a2-macroglobulin. These assays are therefore used mainly for corroborative purposes.
The introduction of heparin in the heparin cofactor assays speeds up the reaction rates and makes them more specific and practical. Antithrombin is often determined by measuring its activity in the presence of an excess of thrombin. Although, heparin cofactor assays that use human thrombin as the target protease may overestimate the antithrombin concentration due to heparin cofactor II; a thrombin-specific serpin with heparin cofactor abilities. It is present in plasma at a concentration about half that of antithrombin and, in the presence of high heparin levels, may represent up to 20% of the thrombin inhibitory activity in plasma. Interference from heparin cofactor II is therefore particulary apparent when analyzing plasma from heparin treated patients. The use of bovine thrombin, low heparin concentrations or factor Xa as a substrate instead of thrombin, minimizes the influence.
Chromogenic heparin cofactor assays
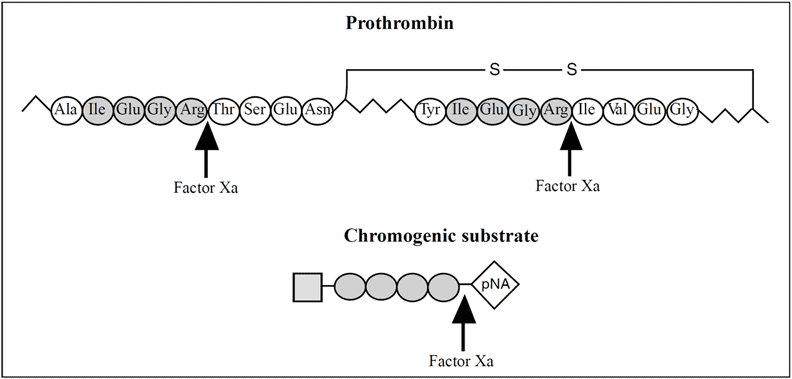
The most frequently applied routine method for measuring antithrombin activity is a heparin cofactor assay based on chromogenic peptide substrates. These mimic either the cleavage site of fibrinogen cleaved by thrombin, or prothrombin cleaved by factor Xa. Considering the influence by heparin cofactor II on assays based on thrombin inhibition, one can expect the chromogenic factor Xa-based assays to completely dominate in the near future.
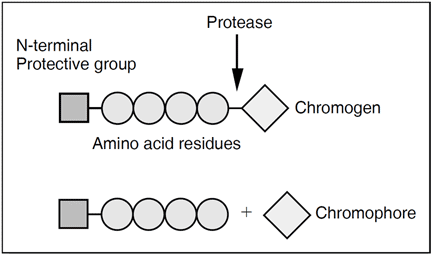
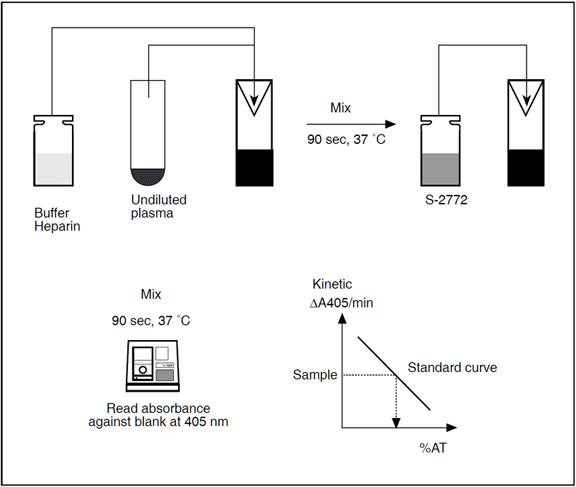
Chromogenic peptides are composed of 3-4 amino acids, with the chromogenic group 4-nitroaniline (pNA) attached to the end. When the peptide is incubated with the active protease it is cleaved and pNA (yellow color) is liberated. The release is measured at 405 nm, either during the reaction in a photometer cuvette, or discontinuously by stopping the reaction with acetic or citric acid. The photometric signal, i.e. the change in absorbance, is inversely proportional to the antithrombin activity.
- Chromogenix S-2222™ Bz-Ile-Glu-(γ-OR)-Gly-Arg-pNA
- Chromogenix S-2765™ Z-D-Arg-Gly-Arg-pNA
- Chromogenix S-2772™ Ac-D-Arg-Gly-Arg-pNA
- Chromogenix S-2238™ H-D-Phe-Pip-Arg-pNA
Chromogenic substrates for factor Xa (Chromogenix S-2222™, Chromogenix S-2765™, Chromogenix S-2772™) and thrombin (Chromogenix S-2238™). Abbreviations: Bz = benzoyl, Z = benzyloxycarbonyl, Ac= acetyl, Pip = pipecolinic acid, Arg = Arginine, Glu = Glutamic acid, Gly = Glycine, Ile = Isoleucine, Phe = Phenylalanine, pNA = para-nitroaniline. Chromogenix S-2222™; R= H (50%) and CH3 (50%).
Coagulometric heparin cofactor assays
As the name implies, antithrombin clotting assays are based on the ability of antithrombin to prolong the clotting time of blood and plasma.
Clotting assays are dependent on manipulation of the plasma sample (defibrination), using prolonged heat treatment or ancrod. Defibrinated plasma is incubated with a fixed amount of thrombin in excess and heparin is added to accelerate the inactivation reaction. This mixture is then added to a fibrinogen solution, and the clotting time, which is directly proportional to the antithrombin activity, is measured. In some assays, factor Xa is used instead of thrombin.
Although clotting assays can be used to measure antithrombin activity accurately, chromogenic assays are preferable as they are more rapid and do not require a defibrination step.
Immunological assays
Immunological antithrombin assays such as the radial immunodiffusion method of Mancini and the electroimmunodiffusion method of Laurell, quantitate the amount of antithrombin antigen present in plasma. The molar concentration of antithrombin protein material in normal plasma, determined using these methods, is approximately 4 mmol/l.
An immunological method in combination with a chromogenic method can be used to diagnose and classify the deficiency states. If both antigen and activity levels are decreased, a quantitative (type I) deficiency can be assumed. They can also provide important information about the consumption of antithrombin in connection with a thrombotic event.
Products
Diagnostic kits from Chromogenix
The development of the first chromogenic peptide substrate S-2160™ in the early Seventies, initiated the introduction of photometry in hematology. Today, there are many tests based on chromogenic substrates for coagulation factors. These tests can be performed manually or on automated analytical systems with high specificity, sensitivity and accuracy.
Important, non-chromogenic-based products include kits for APC resistance (APTT-test).
Antithrombin kits
Chromogenix has well-documented chromogenic test kits for the determination of antithrombin activity in human plasma. The first commercial kit developed was Coatest® Antithrombin (discontinued), which over the years became an established assay worldwide. The Coamatic® Antithrombin kits are the latest development in a line of chromogenic products. A major advantage is that they are based on factor Xa instead of thrombin, thereby eliminating the risk of overestimating the antithrombin level due to the influence of heparin cofactor II. All kits offer a broad measuring range and good reagent stability.
Chromogenix Coamatic® Antithrombin
Chromogenix Coamatic® Antithrombin is a chromogenic heparin cofactor assay for the specific determination of antithrombin activity in plasma. The use of factor Xa eliminates interference from heparin cofactor II and allows accurate antithrombin determination in patients receiving heparin therapy. The assay also discriminates better between antithrombin- deficient and non-antithrombin-deficient individuals compared to a conventional thrombinbased assay. The kit is adapted for use with a wide range of automatic analyzers, achieving comparable reaction conditions on the different analyzers.
Measurement principle
Diluted plasma is incubated with an excess of factor Xa in the presence of heparin. The residual quantity of factor Xa is determined by the rate of hydrolysis of the chromogenic substrate S-2765™. The pNA release measured at 405 nm is inversely proportional to the antithrombin level in the range 0-125% of normal plasma.
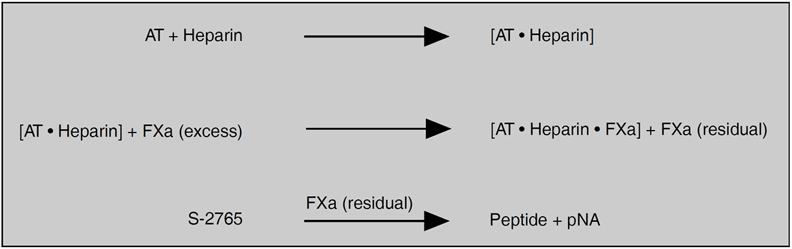
Specificity and interfering factors
No influence from heparin cofactor II, a2-macroglobulin or trypsin inhibitor. No drug interference reported.
Contact activation of plasma may result in the underestimation of antithrombin. Bilirubin, hemoglobin and plasma from hyperlipaemic patients interfere in absorbance readings in the stopped method. In these cases individual plasma blanks are necessary.
Chromogenix Coamatic® LR Antithrombin
Chromogenix Coamatic® LR (liquid reagents) Antithrombin is a chromogenic heparin cofactor assay for the determination of antithrombin activity in undiluted plasma. The reagents of the kit are both in liquid formulation. The use of factor Xa eliminates interference from heparin cofactor II and allows accurate antithrombin determination in patients receiving heparin therapy.
Measurement principle
Plasma is incubated with an excess of factor Xa in the presence of heparin. The residual activity of factor Xa is determined by the rate of hydrolysis of the chromogenic substrate S-2772™. The pNA release measured at 405 nm is inversely proportional to the antithrombin level in the range 0-125% of normal plasma.
Specificity and interfering factors
No influence from heparin cofactor II. No drug interference reported.
Antithrombin reagents
- Chromogenix Antithrombin 10IU
- Chromogenix Antithrombin 25IU