Technozym® Protein C INH Actibind®
$0.00
Technozym® Protein C INH Actibind® is an ELISA kit for determination of active Protein C Inhibitor antigen. The assay is based on the immobilization of functionally active urokinase to plates by means of a monoclonal antibody. Protein C Inhibitor contained in the test sample binds to u-PA and is then quantified using a peroxidase-labeled monoclonal anti-Protein C Inhibitor antibody. The Actibind® PCI can be used as a quantitative assay for the determination of active antigen levels of PCI in subjects with disseminated intravascular coagulation and in atheriosclerotic research.
Determinations: 42 samples in duplicate
| Microtitre Plate | 12 x 8 well plastic microtitre strips precoated with monoclonal anti urokinase coating antibody. |
|---|---|
| Standard | 1 x lyophilized pooled human plasma |
| Urokinase | 1 x lyophilized urokinase 800U/vial |
| POX-Antibody | 1 x conjugated monoclonal anti PCI antibody (concentrated), blue color |
| Sample Dilution Buffer – (white cap) | 2 x 20 ml 2.5-fold concentrated PBS with BSA |
| POX Dilution Buffer – (white cap) | 1 x 12 mL PBS with BSA. |
| Substrate – (green cap) | 1 x 12 ml TMB (Tetramethylbenzidine) in substrate buffer containing H2O2. Ready to use. |
| Stop Solution – (red cap) | 1 x 15 ml 0.5 M Sulphuric Acid. Ready to use. |
| Wash Buffer – (blue cap) | 1 x 20 ml 12.5-fold concentrated PBS with Tween 20 |
| PCI Actibind® High Control | 1 x lyophilized pooled human plasma for lot-specific concentration see label on vial |
Additional Materials (required but not supplied)
- Micropipettes and a multichannel micropipette or multistepper pipette, pipette tips
- Glass or plastic test tubes for diluting the samples.
- Laboratory bottles or beakers and graduated cylinders for diluting wash and dilution buffer
- Distilled or deionised water
- Absorbent paper towels
- Microtitre plate washer (alternatively, washing can be performed manually using a multichannel pipette)
- Microtitre reader equipped with a 450 nm filter and, if possible, a 620 nm reference filter
- Incubator (37 °C)
Measurement Principle
The Technozym® Actibind® PCI test is a solid phase enzyme immunoassay in which an anti-u-PA monoclonal antibody that does not interfere with the active site on the urokinase antigen molecule is coated onto a plastic microtitre plate. Urokinase is incubated on the plate leaving its active site accesible for complex formation with active PCI contained in the sample. Following the binding of the sample the plate is washed and enzyme-labelled (POX = horseradish peroxidase) monoclonal anti-PCI which recognizes another site on the active PCI antigen is incubated on the plate. The quantity of POX which binds is proportional to the quantity of active PCI antigen i.e. non-complexed antigen contained in test samples. Unbound POX-Ab is washed away and a substrate which reacts with the peroxidase enzyme is added, leading to a color change proportional to the amount of enzyme bound. The enzyme reaction is stopped after a specific incubation time. The absorbances of the wells are then measured and the values obtained are used to construct a standard curve from which sample values can be extrapolated.
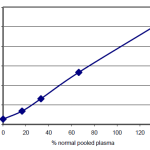
Standard Curve
See Chart
Background
Protein C Inhibitor (PCI) is a member of the serine protease inhibitor (Serpin) superfamily with homology to alpha-1-antichymotrypsin, alpha-1-antitrypsin, antithrombin III, ovalbumin and angiotensinogen. PCI with an apparent molecular weight of 57 KD has been described in a variety of biological fluids including blood plasma (4µg/ml), urine (250ng/ml) and seminal plasma (200µg/ml). Glycosaminoglycan (GAG) dependent PCI inhibits activated protein C (APC), twochain urokinase (u-PA), two-chain tissue plasminogen activator (t-PA), thrombin, factor Xa, and factor XIa in reactions stimulated by heparin. However it has recently been shown that the PCI-tissue kallikrein interaction is inhibited by heparin. Glycosaminogylcans may therefore regulate the enzyme specificity of PCI. PCI inhibits its target proteases by forming SDS stable 1:1 complexes. Upon complex formation the reactive site peptide bond of the inhibitor is cleaved by the protease and the carboxy-terminal peptide is released from the inhibitor. Depending on the target protease, complexes dissociate more or less slowly and cleaved inactive PCI (Mr=54,000) and active enzyme are released.
Although PCI has been shown to inhibit several enzymes, its precise physiological role has yet to be defined. However, the fact that PCI has been determined in high concentrations in a variety of biological fluids indicates its importance as a physiological serine protease inhibitor whether it be as an inhibitor of a specific serine protease in vivo or as a general inhibitor functioning to protect tissues from protease action.
Low values of both Protein C antigen and functional activity of PCI have been determined in subjects with disseminated intravascular coagulation and low antigen values in liver disease and high PCI activity in subjects of MI. uPA-PCI complexes are formed in subjects undergoing thrombolytic testing with u-PA when the concentration of u-PA exceeds the inhibitory capacity of plasminogen activator inhibitor-1 (PAI-1), the primary inhibitor of u-PA in vivo.