Technoview® Apixaban Calibrator Set
$0.00
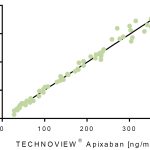
Technoview® apixaban calibrator set is a set of plasma-derived calibration samples with apixaban* concentrations ranging from ~0-500 ng / mL. The set can be used for establishing a standard curve in research measurements of apixiban. It is optimized for use with anti-Xa chromogenic assays and is frequently used in conjunction with the Technoview® apixaban low and high controls in the Technochrom® anti-Xa chromogenic assay.
*Apixaban is the active substance in Eliquis®.
| Calibrator 1 | 1 x 1 mL Technoview Apixaban Calibrator 1 (CAL 1), human plasma, lyophilized, no Apixaban |
| Calibrator 2 | 1 x 1 mL Technoview Apixaban Calibrator 2 (CAL 2), human plasma, lyophilized, ~50 ng / mL Apixaban |
| Calibrator 3 | 1 x 1 mL Technoview Apixaban Calibrator 3 (CAL 3), human plasma, lyophilized, ~180 ng / mL Apixaban |
| Calibrator 4 | 1 x 1 mL Technoview Apixaban Calibrator 4 (CAL 4), human plasma, lyophilized, ~350 ng / mL Apixaban |
| Calibrator 5 | 1 x 1 mL Technoview Apixaban Calibrator 5 (CAL 5), human plasma, lyophilized, ~500 ng / mL Apixaban |
Stability after reconstitution: 18-25°C (open vial) / 48 hours, 2-8°C (closed vial) / 7 days
Advantages
- Allows for establishment of calibration curve covering a range of 0-500 ng/ml
- Optimized for use with anti-Xa (without exogeneous antithrombin) research assays but can validated for use in prothrombin time (PT) research assays
Calibrator Set Composition
5 x 1 ml vials with a range of apixaban concentrations:
- Cal 1: 0 ng/ml
- Cal 2: 50 ng/ml
- Cal 3: 180 ng/ml
- Cal 4: 350 ng/ml
- Cal 5: 500 ng/ml
Calibrators are lyophilized and prepared from pooled human citrated plasma supplemented with different concentrations of apixaban. The plasma contains stabilizers but no bactericide additives. It is calibrated, using an anti-Xa assay, in ng/ml against an internal reference standard and in IU/ml against the international standard for low molecular weight heparin (LMWH) in plasma.
Background
Apixaban (Eliquis®) is an oral direct factor Xa inhibitor drug. It is prescribed by a healthcare provider and can:
- reduce the risk of blood clots and stroke in individuals who have nonvalvular atrial fibrillation;
- treat deep vein thrombosis (DVT) and pulmonary embolism (PE); and
- reduce the risk of DVT and PE in individuals after hip or knee replacement surgery.
In most cases, apixaban does not require routine monitoring. It typically prolongs clotting tests such as PT/INR and aPTT so, if necessary, it is best measured using an anti-Xa chromogenic assay. Note that this calibrator set is for research use only in the US and Canada.
Read more…