
About Peviva
Peviva is a brand of cell death (apoptosis and/or necrosis) ELISAs and antibodies distributed in the US and Canada by DiaPharma. Peviva is now owned by VLVbio.

About VLVbio

VLVbio was established in 2013 in Stockholm Sweden, having been granted exclusive rights to a unique apoptosis marker for keratin 18 (K18) expressing cells of epithelial origin. VLVbio strives to continuously acquire and develop novel intellectual property from the academic research and healthcare market for the detection and quantification of different forms of cell death in normal or pathophysiological circumstances.
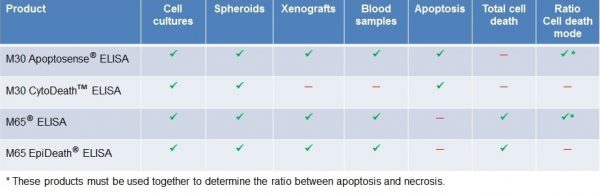
VLVbio ELISAs




VLVbio‘s Corporate Mission
- Invent, develop and market innovative high-quality biomarkers for use in cell-based research in academia, biotech/pharmacy industry and healthcare diagnostics and to expand the product line into related areas of research and diagnostics of different human malignancies.
- Complement these markets through the development of innovative detection and screening technologies.
- Support the advancement of sciences by establishing co-operations with the worldwide community of clinical research as well in partnership with other equally highly committed commercial partner companies.
VLVbio‘s Quality Commitment
VLVbio is committed to providing the highest quality service.
VLVbio’s quality assurance system is certified according to SS-EN ISO 9001 and SS-EN ISO 13485.
The M30 Apoptosense® ELISA, M65®ELISA and M65 EpiDeath® ELISA are registered with the Swedish Medical Products Agency in accordance with the regulations on medical devices and on medical devices for in vitro diagnostics respectively (CE-IVD).
Having produced and delivered thousands of M30 Apoptosense® ELISA and M65® ELISA kits over several years, the quality and reliability of Peviva products have been appreciated and acknowledged by third party publications.




