VACCINE-INDUCED THROMBOTIC THROMBOCYTOPENIA (VITT): INSIGHTS FROM ISTH 2022
Posted on: September 26, 2022
– Contributed by Abi Kasberg, PhD
Vaccine-induced immune thrombotic thrombocytopenia (VITT) is a rare and severe complication of adenoviral-based COVID-19 vaccinations. VITT occurs at a rate of 1:100,000 to 1:1,000,000 and is associated with high morbidity and mortality. Long term complications of VITT and VITT antibodies can persist for more than 9 months and has been termed “long VITT” (Arnold 2022, Gabarin et al. 2022). Characteristics of VITT are similar to heparin-induced thrombocytopenia (HIT), such that disease-causing antibodies against platelet factor 4 (PF4) mediate thrombosis and thrombocytopenia. A key distinguishing feature that separates VITT from HIT is that VITT requires a prior inoculation with adenoviral COVID-19 vaccinations without exposure to heparin. VITT is not associated with underlying thrombotic risk. There are two COVID-19 vaccines most likely to be associated with VITT, the Johnson & Johnson (J&J) / Janssen (Ad26.COV2.S) adenoviral vaccine and the AstraZeneca (ChAdOx1-S) adenoviral vaccine.
It is important to note that as enhanced criteria and testing strategies are being developed to better define and identify VITT, more cases of VITT are being detected with vaccine delivery strategies that are not utilizing adenoviral vectors. A 2021 report indicates that a patient experienced VITT-like symptoms after receiving an inoculation of the Moderna mRNA COVID-19 vaccine (mRNA-1273) (Pishko and Cuker 2021, Sangli et al. 2021). Additionally, 32 cases of major thrombosis with thrombocytopenia have been reported to the Medicines and Healthcare products Regulatory Agency (MHRA) in the UK following Pfizer mRNA COVID-19 vaccine (BNT162b2), along with 8 reported cases following the Moderna COVID-19 vaccine, as of August 2022. Although VITT is commonly thought to be specific to adenoviral-based COVID-19 vaccines, further studies need to be performed to determine the potential association of VITT outcomes with additional vaccine delivery types. Thus, it is necessary to have accurate ways of diagnosing and researching the underlying mechanisms of VITT as new vaccines are developed. When considering vaccine development in non-infectious disease areas, like oncology, the impact of various vaccine technologies could become a far-reaching area of interest down the line.
A conclusive VITT diagnosis must include the following criteria:
|
The mechanism of VITT pathogenesis is an area of ongoing investigation. What has been understood thus far is that VITT immunoglobulin G (IgG) antibodies recognize the chemokine PF4. PF4 is released from the α-granules of platelets. FcγIIa receptors on the platelet surface bind the low-affinity Fc domain of IgG in a mechanism that activates platelets (Fig. 1). Platelet activation stimulates the coagulation pathway, enabling further downstream thrombotic complications to occur. VITT antibodies also stimulate tissue factor (TF) expression on monocytes, inducing inflammation (Fig. 1) (Iba et al. 2022). Following innate immune system activation, neutrophils release neutrophil extracellular traps (NETs) that contribute to heightened coagulation cascades (Fig. 1) (Iba et al. 2022).
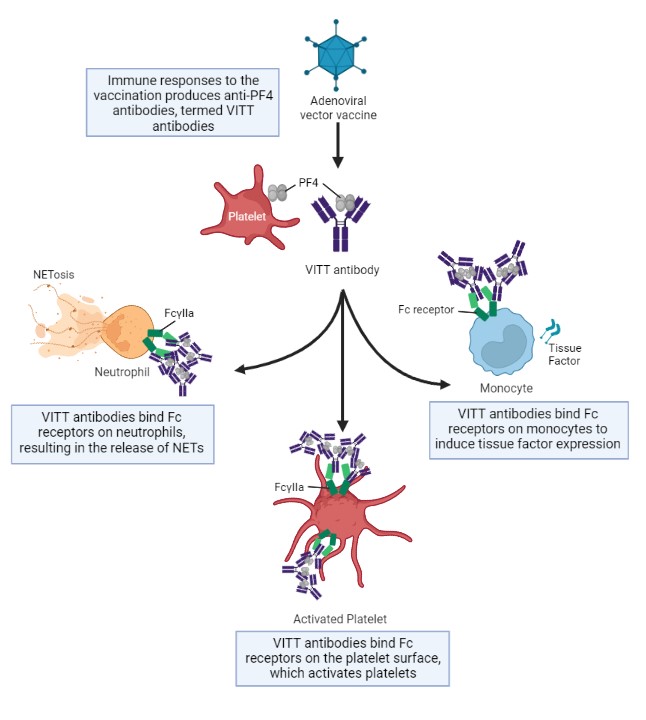
Figure 1: Proposed mechanisms of VITT pathogenesis that contribute to platelet activation, stimulation of the coagulation cascade, inflammation, and ultimately thrombosis. Neutrophil extracellular traps (NETs); platelet factor 4 (PF4); vaccine-induced immune thrombotic thrombocytopenia (VITT). Figure created with BioRender.com
A prominent mystery surrounding VITT is the initial trigger. The J&J and AstraZeneca COVID-19 vaccines are both adenoviral vector-based, suggesting that perhaps a vector-specific mechanism may be driving VITT pathogenesis (Iba et al. 2022). In support of the hypothesis that the adenoviral vector may be contributing to VITT, surface plasmon resonance technology has recorded the formation of stable complexes of adenoviruses, including ChAdOx1, to PF4 (Baker et al. 2021). PF4/vaccine neoantigens in combination with vaccine-induced inflammation stimulates B cells to generate anti-PF4 antibodies that drive the pathogenic outcomes observed in VITT, including the release of NETs (Greinacher et al. 2021). On the contrary, adenoviral vectors have historically been used to create Ebola and AIDS vaccines without VITT-like adverse events being reported (Iba et al. 2022).
A current hypothesis suggests that VITT may be similar to autoimmune HIT in that a strong immune response may be triggered by contents within the vaccine preparation, such as EDTA or human proteins in the vaccine (Greinacher et al. 2021, McGonagle et al. 2021). To investigate this, additional immune factors should be analyzed such as neutrophils, NETs, monocyte activation, inflammasome complexes, and endothelial cell activation (Kashir et al. 2021). Vaccine preparation impurities, contaminants, or differences in purity between vaccine lots may be triggering VITT (Krutzke et al. 2022). There is a need for further investigation into the cause and pathogenesis of VITT in order to better understand the unusual incidence of abdominal and cerebral thrombosis (Iba et al. 2022).
Clinical treatment regimens for VITT are still being established. In vitro studies have suggested that VITT can be treated with intravenous immunoglobulin (IVIG) therapy or with a monoclonal antibody that blocks the Fc receptor to prevent platelet activation by VITT antibodies (Gabarin et al. 2022). Non-heparin anticoagulants such as direct thrombin inhibitors, Factor Xa inhibitors, and direct oral anticoagulants (DOACs) have been suggested for use (Gabarin et al. 2022). Heparin and danaparoid anticoagulants have also shown to inhibit thrombus formation and suppress platelet activation (Arnold 2022, Tiede et al. 2021). It has been suggested that heparin binds PF4 at the same amino acid location as VITT antibodies (Huynh et al. 2021). Therefore, heparin treatment dissociates the PF4/VITT antibody complex to mitigate VITT (Arnold 2022). Due to the newness of VITT and the global utilization of adenoviral vector vaccines worldwide, there are many urgent clinical needs and research questions that require attention.
ISTH 2022 VITT Summary
Insightful and exceptional research presentations were recently given at the International Society on Thrombosis and Haemostasis (ISTH) Congress 2022 hosted in London, England, UK. Of interest, talks reported on the status, urgency, and obstacles encountered during VITT research. Here, is a review of some key VITT research talks that were presented at ISTH 2022.
Distinguishing VITT from HIT, Anand Padmanabhan, MBBS, PhD
A major roadblock to the investigation of VITT is the inability to differentiate between HIT, spontaneous HIT, and VITT via standard laboratory tests. HIT ELISA tests detect anti-PF4 antibodies in VITT and HIT samples, but other HIT assays do not work reliably at detecting VITT. In fact, commercially available antigen and functional assays, including HIT ELISA tests, are unable to differentiate between VITT, HIT, and spontaneous HIT. At ISTH 2022, Dr. Anand Padmanabhan described how functional serotonin release assays (SRA) that utilize heparin-treated platelets have low-sensitivity and low specificity for detecting pathological VITT antibodies. Functional assays utilizing PF4-treated platelets, such as the P-selectin expression assay (PEA) and the PF4-induced platelet activation (PIPA) test, are sensitive and accurate for detecting VITT antibodies. However, these functional assays are complex to execute and often require specialized reference laboratory services with limited accessibility (Kanack et al. 2022C).Likewise, antigenic assays to detect PF4-polyanion complexes show high-sensitivity but low specificity in VITT detection. These current diagnostic HIT detection methods are therefore inadequate for detection of VITT antibodies.
Antigenic immunoassays that identify anti-PF4/heparin antibodies are frequently used to detect initial immune responses during HIT pathogenesis (Sahu et al. 2020). The accurate diagnosis and management of VITT may likewise depend on anti-PF4 antibody immunoassays. To circumvent the low specificity observed when utilizing traditional HIT ELISAs, Dr. Padmanabhan described the utilization of un-complexed PF4 ELISA assays to detect VITT antibodies (Kanack et al. 2022A). Un-complexed PF4 ELISAs do not utilize the PF4/polyanion complex to detect VITT antibodies. Dr. Padmanabhan’s results showed that un-complexed PF4 ELISAs display 100% sensitivity and 100% specificity at detecting VITT antibodies in small sample sizes (n=6) (Kanack et al. 2022A). This suggests that VITT antibody binding to un-complexed PF4 is a key diagnostic feature of VITT.
To explore the underlying complications arising from non-adenoviral-based vaccinations, Dr. Padmanabhan presented on a patient case of thrombosis with thrombocytopenia syndrome (TTS) following administration of the Moderna mRNA COVID-19 vaccine (See et al. 2022). Patient data generated from the un-complexed PF4 ELISA assay indicates the presence of VITT antibodies (Padmanabhan et al. 2022). This suggests the possibility that the TTS event observed in this case involving a non-adenoviral, mRNA-based vaccine was caused by VITT (Padmanabhan et al. 2022, See et al. 2022). In addition, un-complexed PF4 ELISA data from a study of GARDASIL®9 human papillomavirus 9-valent (HPV) vaccine that utilizes virus-like particle (VLP) technology suggests VITT may be the cause of reported adverse events (Kanack et al. 2022B, Johansen et al. 2021).
Together this novel research suggests that un-complexed PF4 ELISAs may be more specific for the detection of VITT than traditionally used HIT PF4/polyanion ELISAs (Fig. 2). Due to limited sample sizes, more research is needed to investigate the specificity of un-complexed PF4 ELISAs to distinguish VITT from spontaneous HIT.
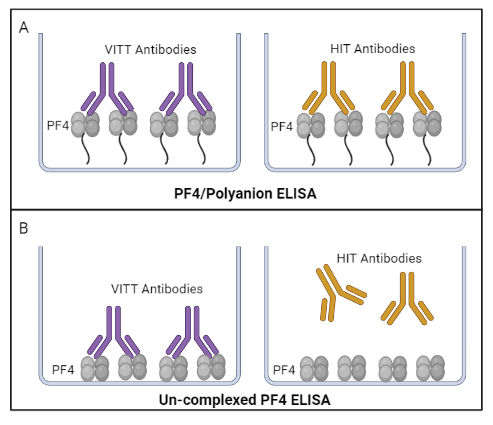
Figure 2: (A) VITT antibodies and HIT antibodies bind PF4/polyanion ELISA assays. (B) VITT antibodies bind un-complexed PF4 ELISAs with high specificity and sensitivity, unlike HIT antibodies. Heparin-induced thrombocytopenia (HIT); platelet factor 4 (PF4); vaccine-induced immune thrombotic thrombocytopenia (VITT). Figure created with BioRender.com
A new antigenic target of VITT antibodies, Conroy Field, MS
It has been postulated that VITT is likely mediated through platelet-activating antibodies that bind PF4 (Huynh et al. 2021). However, it is also possible that additional serum factors or antibodies may be contributing to thrombotic events in VITT (Huynh et al. 2021). When comparing VITT antibodies to HIT antibodies, the antigenic sites for VITT antibodies that bind PF4 differ from the HIT antibody antigenic sites for PF4. VITT antibody binding sites instead overlap with heparin binding sites on PF4, such that VITT antibodies mirror the effects of heparin on PF4 (Huynh et al. 2021). This is a fascinating characteristic to consider due to the therapeutic potential of leveraging heparin to block VITT antibody binding to PF4 (Huynh et al. 2021). It has not been determined if VITT antibodies cross-react with heparin-PF4 complexes. This is an important component to investigate when considering the therapeutic potential of heparin as an anticoagulant in VITT.
During a great presentation at ISTH 2022, Conroy Field described that VITT antibodies have a highly conserved binding site to neutrophil-activating peptide 2 (NAP2), suggesting that NAP2 could be an additional antigenic target in VITT. NAP2 and PF4 have similarities in that they both are platelet-specific chemokines, are positively charged proteins, bind heparin, are stored in platelet α-granules, and released in high amounts upon platelet activation (Witt and Lander 1994). NAP2 is unique from PF4 in that NAP2 is an N-terminal cleavage product from platelet basic protein (PBP) and functions as a neutrophil agonist via CXCR2 chemokine receptor (Zlotnik and Yoshie 2012).
In vitro and mouse model studies were performed to investigate the role that NAP2 has during VITT pathogenesis. In vitro studies show that VITT antibodies bind both PF4 and NAP2 in mechanisms that facilitate platelet activation. Murine studies using a transgenic FcγRIIa/Pf4-/- mouse model for VITT further suggested that VITT antibodies (VITT IgG) induce a prothrombotic state in the absence of PF4. This suggests that PF4 is not required for thrombotic states in VITT and that another chemokine may be contributing to thrombosis in VITT.
To further investigate the role that neutrophils play in VITT, neutrophil velocity was recorded in FcγRIIa/Pf4-/- mutant mice. In the absence of PF4, neutrophil velocity slows down and becomes sticky following administration of VITT IgG. Further supporting studies showed that VITT antibodies do not induce thrombosis in the absence of both Pf4 and Nap2. Following VITT IgG induction, neutrophils and platelets accumulated in venous and aterial thrombi. This contrasts with HIT, where neutrophils do not accumulate in arterial thrombi (Gollomp et al. 2018).
Together, this fascinating research suggests that VITT antibodies target and bind both PF4 and NAP2 to activate platelets (Fig. 3) (Rauova et al. 2021). This causes an accumulation of platelets and neutrophils in arterial and venous thrombi in a mechanism that appears to be unique to VITT. The proposed involvement of neutrophils in VITT thrombosis could be initiated through VITT antibody binding on the neutrophil agonist NAP2 (Fig. 3). More research is needed regarding the role of NAP2-mediated activation of neutrophils during immunothrombotic responses in VITT (Rauova et al. 2021). This suggests that anti-NAP2 VITT antibodies are influencing the pathogenesis of VITT and therefore may be novel therapeutic targets to consider.
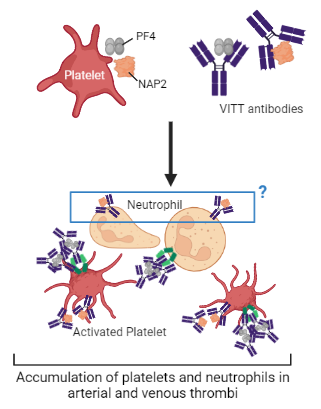
Figure 3: VITT antibodies may be targeting NAP2 in addition to PF4 to activate platelets. It remains unknown whether NAP2, in the company of VITT antibodies, functions to mediate neutrophil activation during VITT pathogenesis, as outlined in the blue box. Neutrophil-activating peptide 2 (NAP2), platelet factor 4 (PF4), vaccine-induced immune thrombotic thrombocytopenia (VITT). Figure created with BioRender.com
Further Reading
Arnold DM. Heparin or nonheparin anticoagulants for VITT. Blood. 2022 Jun 9;139(23):3358-3359. doi: 10.1182/blood.2022016423. PMID: 35679072; PMCID: PMC9181564.
Baker AT, Boyd RJ, Sarkar D, Teijeira-Crespo A, Chan CK, Bates E, Waraich K, Vant J, Wilson E, Truong CD, Lipka-Lloyd M, Fromme P, Vermaas J, Williams D, Machiesky L, Heurich M, Nagalo BM, Coughlan L, Umlauf S, Chiu PL, Rizkallah PJ, Cohen TS, Parker AL, Singharoy A, Borad MJ. ChAdOx1 interacts with CAR and PF4 with implications for thrombosis with thrombocytopenia syndrome. Sci Adv. 2021 Dec 3;7(49):eabl8213. doi: 10.1126/sciadv.abl8213. Epub 2021 Dec 1. PMID: 34851659; PMCID: PMC8635433.
Gabarin N, Arnold DM, Nazy I, Warkentin TE. Treatment of vaccine-induced immune thrombotic thrombocytopenia (VITT). Semin Hematol. 2022 Apr;59(2):89-96. doi: 10.1053/j.seminhematol.2022.03.002. Epub 2022 Mar 7. PMID: 35512906; PMCID: PMC8898785.
Gollomp K, Kim M, Johnston I, Hayes V, Welsh J, Arepally GM, Kahn M, Lambert MP, Cuker A, Cines DB, Rauova L, Kowalska MA, Poncz M. Neutrophil accumulation and NET release contribute to thrombosis in HIT. JCI Insight. 2018 Sep 20;3(18):e99445. doi: 10.1172/jci.insight.99445. PMID: 30232279; PMCID: PMC6237233.
Greinacher A, Selleng K, Palankar R, Wesche J, Handtke S, Wolff M, Aurich K, Lalk M, Methling K, Völker U, Hentschker C, Michalik S, Steil L, Reder A, Schönborn L, Beer M, Franzke K, Büttner A, Fehse B, Stavrou EX, Rangaswamy C, Mailer RK, Englert H, Frye M, Thiele T, Kochanek S, Krutzke L, Siegerist F, Endlich N, Warkentin TE, Renné T. Insights in ChAdOx1 nCoV-19 vaccine-induced immune thrombotic thrombocytopenia. Blood. 2021 Dec 2;138(22):2256-2268. doi: 10.1182/blood.2021013231. PMID: 34587242; PMCID: PMC8483989.
Huynh A, Kelton JG, Arnold DM, Daka M, Nazy I. Antibody epitopes in vaccine-induced immune thrombotic thrombocytopaenia. Nature. 2021 Aug;596(7873):565-569. doi: 10.1038/s41586-021-03744-4. Epub 2021 Jul 7. PMID: 34233346.
Iba T, Levy JH, Warkentin TE. Recognizing Vaccine-Induced Immune Thrombotic Thrombocytopenia. Crit Care Med. 2022 Jan 1;50(1):e80-e86. doi: 10.1097/CCM.0000000000005211. PMID: 34259661; PMCID: PMC8670081.
Johansen S, Laegreid IJ, Ernstsen SL, Azrakhsh NA, Kittang AO, Lindås R, Gjertsen BT, Vetti N, Mørtberg TV, Sørvoll IH, Holme PA, Ahlen MT, Reikvam H. Thrombosis and thrombocytopenia after HPV vaccination. J Thromb Haemost. 2022 Mar;20(3):700-704. doi: 10.1111/jth.15604. Epub 2021 Dec 7. PMID: 34817130.
Kanack AJ, Singh B, George G, Gundabolu K, Koepsell SA, Abou-Ismail MY, Moser KA, Smock KJ, Green D, Major A, Chan CW, Wool GD, Reding M, Ashrani AA, Bayas A, Grill DE, Padmanabhan A. Persistence of Ad26.COV2.S-associated vaccine-induced immune thrombotic thrombocytopenia (VITT) and specific detection of VITT antibodies. Am J Hematol. 2022A May;97(5):519-526. doi: 10.1002/ajh.26488. Epub 2022 Feb 21. PMID: 35132672; PMCID: PMC8986571.Kanack AJ, Laegreid IJ, Johansen S, Reikvam H, Ahlen MT, Padmanabhan A. Human papilloma virus vaccine and VITT antibody induction. Am J Hematol. 2022B Oct;97(10):E363-E364. doi: 10.1002/ajh.26659. Epub 2022 Aug 3. PMID: 35834243; PMCID: PMC9463111.
Kanack AJ, Jones C, Singh B, Leger R, Splinter N, Heikal NM, Pruthi RK, Chen D, George G, Abou-Ismail MY, Wool GD, Gundabolu K, Padmanabhan A. Off-the-shelf cryopreserved platelets for the detection of HIT and VITT antibodies. Blood. 2022C Aug 23:blood.2022017283. doi: 10.1182/blood.2022017283. Epub ahead of print. PMID: 35998675.
Kashir J, Ambia AR, Shafqat A, Sajid MR, AlKattan K, Yaqinuddin A. Scientific premise for the involvement of neutrophil extracellular traps (NETs) in vaccine-induced thrombotic thrombocytopenia (VITT). J Leukoc Biol. 2022 Mar;111(3):725-734. doi: 10.1002/JLB.5COVR0621-320RR. Epub 2021 Sep 1. PMID: 34467562; PMCID: PMC8667645.
Krutzke L, Rösler R, Allmendinger E, Engler T, Wiese S, Kochanek S. Process- and product-related impurities in the ChAdOx1 nCov-19 vaccine. Elife. 2022 Jul 4;11:e78513. doi: 10.7554/eLife.78513. PMID: 35781137; PMCID: PMC9313527.
McGonagle D, De Marco G, Bridgewood C. Mechanisms of Immunothrombosis in Vaccine-Induced Thrombotic Thrombocytopenia (VITT) Compared to Natural SARS-CoV-2 Infection. J Autoimmun. 2021 Jul;121:102662. doi: 10.1016/j.jaut.2021.102662. Epub 2021 May 19. PMID: 34051613; PMCID: PMC8133385.
Padmanabhan A, Kanack AJ, Kaplan RB, Sangli S. COVID-19 mRNA-1273 vaccine induces production of vaccine-induced immune thrombotic thrombocytopenia antibodies. Am J Hematol. 2022 Jun 1;97(6):E223-E225. doi: 10.1002/ajh.26542. Epub 2022 Mar 26. PMID: 35312193; PMCID: PMC9081112.
Pishko AM, Cuker A. Thrombosis After Vaccination With Messenger RNA-1273: Is This Vaccine-Induced Thrombosis and Thrombocytopenia or Thrombosis With Thrombocytopenia Syndrome? Ann Intern Med. 2021 Oct;174(10):1468-1469. doi: 10.7326/M21-2680. Epub 2021 Jun 29. PMID: 34181441; PMCID: PMC8251937.
Rauova L, Wang A, Yarovoi S, Khandelwal S, et al. Vaccine-Induced Thrombocytopenia and Thrombosis (VITT) Antibodies Recognize Neutrophil-Activating Peptide 2 (NAP2) As Well As Platelet Factor 4 (PF4): Mechanistic and Clinical Implications. Blood (2021) 138 (Supplement 1): 292. doi.org/10.1182/blood-2021-151685
Sahu KK, Jindal V, Anderson J, Siddiqui AD, Jaiyesimi IA. Current Perspectives on Diagnostic Assays and Anti-PF4 Antibodies for the Diagnosis of Heparin-Induced Thrombocytopenia. J Blood Med. 2020 Aug 17;11:267-277. doi: 10.2147/JBM.S232648. PMID: 32884385; PMCID: PMC7443028.
Sangli S, Virani A, Cheronis N, Vannatter B, Minich C, Noronha S, Bhagavatula R, Speredelozzi D, Sareen M, Kaplan RB. Thrombosis With Thrombocytopenia After the Messenger RNA-1273 Vaccine. Ann Intern Med. 2021 Oct;174(10):1480-1482. doi: 10.7326/L21-0244. Epub 2021 Jun 29. PMID: 34181446; PMCID: PMC8251935.
See I, Lale A, Marquez P, Streiff MB, Wheeler AP, Tepper NK, Woo EJ, Broder KR, Edwards KM, Gallego R, Geller AI, Jackson KA, Sharma S, Talaat KR, Walter EB, Akpan IJ, Ortel TL, Urrutia VC, Walker SC, Yui JC, Shimabukuro TT, Mba-Jonas A, Su JR, Shay DK. Case Series of Thrombosis With Thrombocytopenia Syndrome After COVID-19 Vaccination-United States, December 2020 to August 2021. Ann Intern Med. 2022 Apr;175(4):513-522. doi: 10.7326/M21-4502. Epub 2022 Jan 18. PMID: 35038274; PMCID: PMC8787833.
Tiede A, Sachs UJ, Czwalinna A, Werwitzke S, Bikker R, Krauss JK, Donnerstag F, Weißenborn K, Höglinger G, Maasoumy B, Wedemeyer H, Ganser A. Prothrombotic immune thrombocytopenia after COVID-19 vaccination. Blood. 2021 Jul 29;138(4):350-353. doi: 10.1182/blood.2021011958. PMID: 34323939; PMCID: PMC8084604.
Witt DP, Lander AD. Differential binding of chemokines to glycosaminoglycan subpopulations. Curr Biol. 1994 May 1;4(5):394-400. doi: 10.1016/s0960-9822(00)00088-9. PMID: 7922353.
Zlotnik A, Yoshie O. The chemokine superfamily revisited. Immunity. 2012 May 25;36(5):705-16. doi: 10.1016/j.immuni.2012.05.008. PMID: 22633458; PMCID: PMC3396424.
ML-00-00973